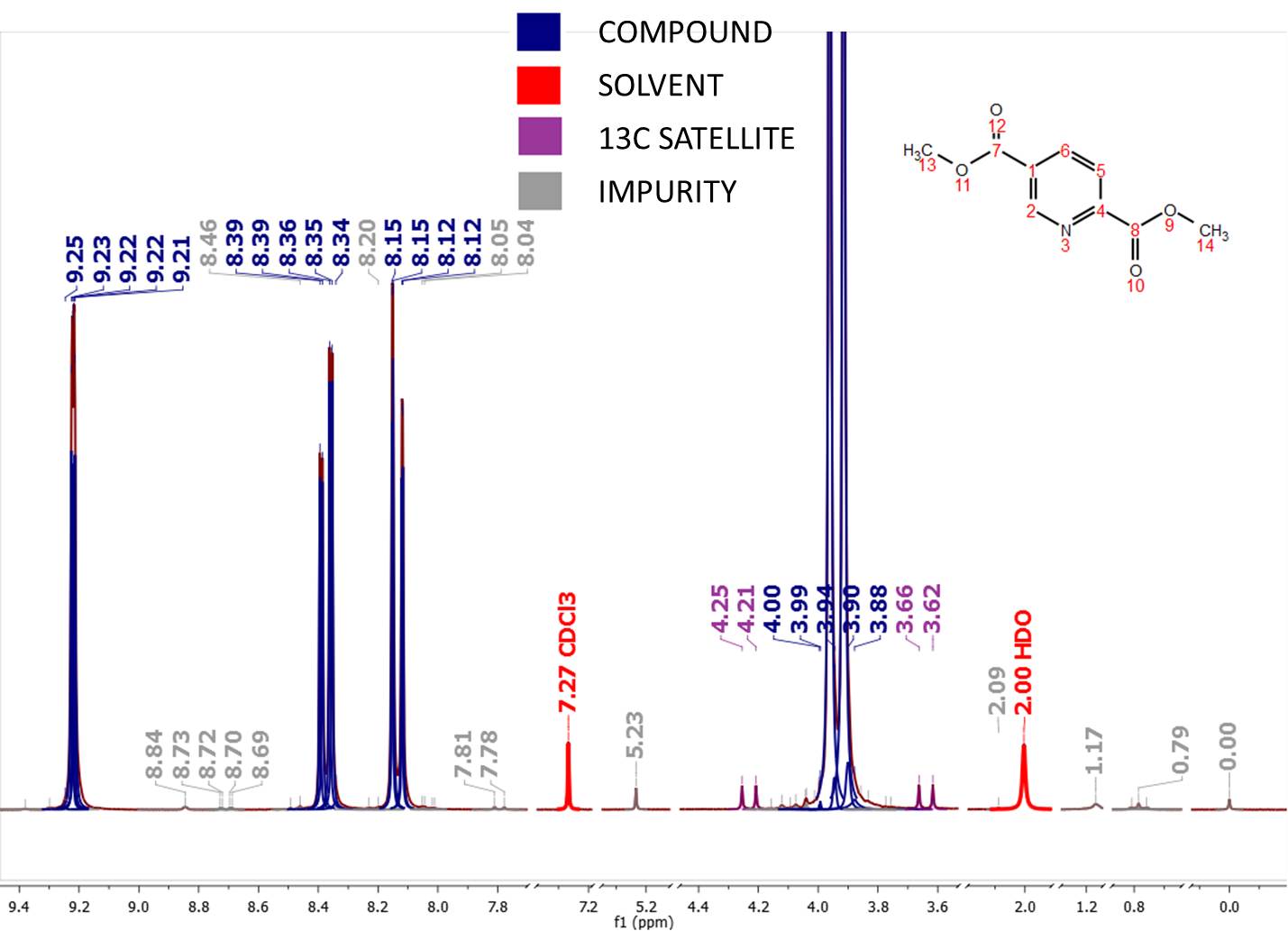
NMR is a quantitative spectroscopic tool because the intensity of the peaks is directly proportional to the number of nuclei. Because of the distinct advantages of each method, NMR and MS are considered as complementary techniques. NMR is less sensitive than MS, which can detect compounds down to parts per million (ppm) levels. Because NMR is able to detect compounds only down to 0.1% level, it is not suitable for the detection of trace components. The standardization of the NMR protocol will further improve the usefulness of NMR as a tool for the profiling of natural products extracts. In contrast, NMR does not require elaborate sample preparation and fractionation, is highly reproducible, and is able to provide both qualitative and quantitative information on chemically diverse compounds. These make it difficult to standardize MS analysis. LC-MS, on the other hand, generally requires sample preparation, chromatographic separation, specific experimental and ionization conditions, instrumentation and operator skill. In particular, NMR is nondestructive, unbiased, quantitative, does not require separation or derivatization, and is amenable to compounds that are difficult to analyze by GC-MS and LC-MS. For example, GC-MS often requires derivatization of compounds, such as sugars and amines. The standardization of the NMR protocol will make it a more useful tool for the profiling of natural products extracts.Īlthough MS techniques, such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS), are most commonly used in metabolomics, NMR still enjoys a number of key advantages. The use of two-dimensional 1H NMR has been used to overcome problems of spectral overlap. Various applications of 1H and 13C NMR for the profiling of natural products are described. Although 1H NMR is more sensitive, 13C NMR spectra are simpler with less overlapping signals and are less affected by different magnetic field strengths. This paper will describe the basic theoretical principles that should be considered to develop NMR into a standard quantitative method. These approaches, however, are not mutually exclusive and will likely converge in the future. There are two general approaches to the use of NMR for profiling studies: an untargeted approach, which uses chemometric analysis and a targeted approach, which aims to quantify known compounds in the extract. Although MS is gaining popularity in metabolomics, NMR enjoys a number of key advantages because it is nondestructive, unbiased, quantitative, does not require separation or derivatization, and is amenable to compounds that are difficult to analyze by gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS). But, vicinal coupling refers to the coupling of two hydrogen atoms that are bound to two adjacent carbon atoms.Nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry (MS) are the principal methods of metabolomics, the branch of ‘-omics’ that deals with small molecules. The key difference between geminal and vicinal coupling is that geminal coupling refers to the coupling of two hydrogen atoms that are bound to the same carbon atom. Thus, the parental gametes are AB and ab. Coupling refers to the case where dominant alleles are on the same homologue chromosome and both recessive alleles are on the other homologue chromosome. The terms coupling and repulsion only make sense if the two genes show complete dominance. Most importantly, J-coupling provides information on the connectivity of chemical bonds. In NMR spectroscopy, J-coupling contains information about relative bond distances and angles. It is an indirect interaction between two nuclear spins that arises from hyperfine interactions between the nuclei and local electrons. The vicinal coupling constant is referred to as 3J because the hydrogen atoms couple through three bonds.Īlso question is, What is coupling in NMR?. In 1H NMR spectroscopy, the coupling of two hydrogen atoms on adjacent carbon atoms is called vicinal coupling. Similarly, it is asked, What is vicinal coupling in NMR spectroscopy? The hydrogen atoms are on the adjacent carbon atoms in vicinal coupling. The vicinal or 3 bond coupling constants is denoted by 3J.
